Pregnancy incidence in patients affected by chronic kidney disease (CKD) is increasing, despite the fact that renal disease is associated with high maternal and fetal morbidity. From an obstetrical perspective, the main complications for the developing pregnancy are often represented by preeclampsia or preterm delivery, in addition to the progression of their underlying renal dysfunction on a continuously increasing physiologic demands. If a pregnancy is considered, preconception counseling is advised. The lower the stage of CKD, the safer it is to consider in conceiving; however, in recent years, pregnancy in advanced or end-stage renal disease (ESRD), while on dialysis, is not a rare event, as overall outcome for those with advanced CKD has improved over time. The final risk of negative maternal-fetal outcomes is inversely related to renal function and increases with proteinuria. A multidisciplinary approach involving obstetricians, nephrologists and also neonatologists is needed in order to improve pregnancy outcomes in women with chronic kidney disease. The frequency of nephrological and obstetrical examination for pregnant CKD patients should be individualized for each patient.
Impactul bolii renale cronice în sarcină asupra mamei şi fătului. Prezentare de două cazuri şi review al literaturii
Adverse maternal and fetal outcomes in pregnant patients with chronic kidney disease. Presentation of two cases in our clinic and literature review
First published: 21 mai 2019
Editorial Group: MEDICHUB MEDIA
DOI: 10.26416/Gine.24.2.2019.2373
Abstract
Rezumat
Incidenţa sarcinii la pacientele cu boală renală cronică (BRC) este în continuă creştere, în ciuda faptului că patologia renală se asociază cu o creştere a morbidităţii materne şi fetale. Din punct de vedere obstetrical, principalele complicaţii ce pot pune în pericol sarcina sunt adesea reprezentate de preeclampsie sau naştere prematură, complicaţii ce apar suplimentar progresiei disfuncţiei renale, suprapusă nevoilor fiziologice în continuă creştere. Atunci când un cuplu ia în considerare obţinerea unei sarcini, se recomandă consiliere preconcepţională. Cu cât stadiul BRC este mai mic, cu atât este mai sigură obţinerea unei sarcini, însă în ultimii ani sarcina obţinută în stadii avansate sau în stadiul terminal al bolii renale cronice (ESRD), în timpul tratamentului de substituţie renală (dializă), nu mai este un eveniment rar, întrucât prognosticul pentru aceste paciente s-a îmbunătăţit de-a lungul timpului. Riscul final al impactului negativ materno-fetal este invers proporţional cu funcţia renală şi creşte cu proteinuria. O abordare multidisciplinară, care implică medicul obstetrician, nefrolog şi, de asemenea, medicul neonatolog, este necesară pentru a îmbunătăţi prognosticul sarcinii la pacientele cu boală renală cronică. Frecvenţa examinării nefrologice şi obstetricale la gravidele cu BRC trebuie individualizată şi adaptată fiecărei paciente.
Introduction
Chronic kidney disease (CKD) is not as uncommon as thought, even though it compromises fertility, as most of the patients on dialysis suffer from amenorrhea or have anovulatory cycles(1). The incidence of renal disease in women in their reproductive years is approximately 3%(2). It has been suggested that early-stage CKD (stages 1-2) is present in 3:100 pregnancies and advanced-stage CKD (stages 3-5) is present in 1:750 pregnancies(3). Intensive hemodialysis, of at least 20 hours per week, has been shown to improve pregnancy outcomes, both for the mother as for the fetus, and it is at the moment a key therapeutic method for young women with advanced CKD and end-stage renal disease (ESRD)(4). There is a strict relationship between the intensity (in manners of frequency and duration) of the dialysis sessions and pregnancy results, thus leading to intensify dialysis up to daily, as we intended with the patients in our clinic, favoring long-hours treatment as compared to standard schedules(5). The ideal time spent on dialysis is 36 hours per week and the target serum urea nitrogen should be under 50 mg/dl(4). A more intense dialysis schedule offers better disposal of uremic toxins that helps establish the natural balance and normal renal physiology and implicitly improve the placental blood flow(6), thus offering a better chance of survival for the fetus.
The start point for dialysis in pregnancy differs from the one in the average patient; while standard treatment outside pregnancy starts at levels of less than 10 mL/min, when considering a pregnant patient, it can be started from 20 mL/min(7), as glomerular filtration rate (GFR) increases physiologically by approximately 50%.
Studies showed that a favorable prognosis for CKD patients is more likely as the level of serum uric acid is lower and also in the absence of hypertension before conceiving, data that can be documented by an adequate prenatal care and periodic medical check-up(8). But this rarely happens, as many of the pregnant patients learn about their renal malfunction only after becoming pregnant, or even worse, in the late second trimester or in the third trimester of pregnancy, when differential diagnosis with preeclampsia is more difficult.
Along with all the biochemical and hemodynamic changes, our attention should also be directed to other aspects of the pregnant patient, such as mental health and well-being, as this disease can have a great emotional impact especially on young women, regarding their chronic disease or the impossibility of giving their child the best start-up in life(9). Factors affecting pregnancy include hypertension, proteinuria and teratogenic medication. It is important to be able to inform before conceiving on the risks that the mother and the baby are exposed to during pregnancy, in order to take the best informed decision(10).
Cases presentation
We present two cases that met the criteria for chronic kidney disease, described in pregnant patients, admitted in the Obstetrics and Gynecology Department of the Bucharest University Emergency Hospital, Romania, during the past 1.5 years. Our objective was to compare our cases and their outcome to existing data and research published so far.
Case 1
The first patient, a 35-year-old Caucasian woman, presented at 31 weeks of pregnancy, previously admitted in the Nephrology Department of Bucharest University Emergency Hospital for chronic kidney disease and uremia, where she was undertaking hemodialysis sessions, by percutaneous catheterization of the internal jugular vein, up to two or three times per week. She was diagnosed, six years prior to this episode, with secondary hypertensive disorder, with a maximum value of 180/110 mmHg, for which she began antihypertensive medication, and she was diagnosed with chronic kidney disease of unknown etiology, a form of CKD that appears unrelated to traditional risk factors, such as hypertension, diabetes or glomerulonephritis and which typically appears in the mid-thirties to forties population. Ever since, she was under antihypertensive medication, with nutritional support for renal disease, and eventually recombinant human erythropoietin associated with iron supplements. She previously had a successful pregnancy and parturition, at the age of 22 years old, with just a urinary tract infection during the third trimester, with no following complications. We also noted general information about lifestyle, heredocolateral history, and personal physiological and pathological history – she denied any toxic environment exposure, alcohol or drug consumption. She was a nurse practitioner for about 15 years. To be noted: the patient had a relative on maternal line, the maternal aunt, with unilateral renal hypoplasia. No other events to be noted.
At the moment of her admission in the Department of Obstetrics and Gynecology of the Bucharest University Emergency Hospital, both the obstetrical and nephrological teams were focused on offering the best prenatal and intrapartum care. We performed daily ultrasound examination and non-stress test, as well as renal function monitoring by daily blood samples. Fetal well-being and fetal growth were assessed by serial measurements and ultrasound biometry, and Doppler velocimetry of uterine and umbilical arteries were assessed; as a result, the diagnose of intrauterine growth restriction and polyhydramnios, with AFI=20, was established. Dexamethasone for induction of lung maturation was previously administered at standard doses. The patient was slightly malnourished, with poor representation of adipose tissue. She had a moderate anemia (hemoglobin was 7.7 g/dl), moderate thrombocytopenia (77 x 103/mL), a mild hypopotassemia (3.1-3.4 mmol/l), proteinuria (3,987.5 mg/24 hours), with a level of albuminuria of 1977 mg/24 hours and nitrogen retention (urea) 220 mg/dl, plus a creatinine level of 9.44 mg/dl.
The variations of hemoglobin, creatinine, uric acid, and urea during the admission in the Department of Obstetrics and Gynecology of the Bucharest University Emergency Hospital are presented in Figure 1. She received the appropriate treatment, including two units of blood transfusions and recombinant human erythropoietin.
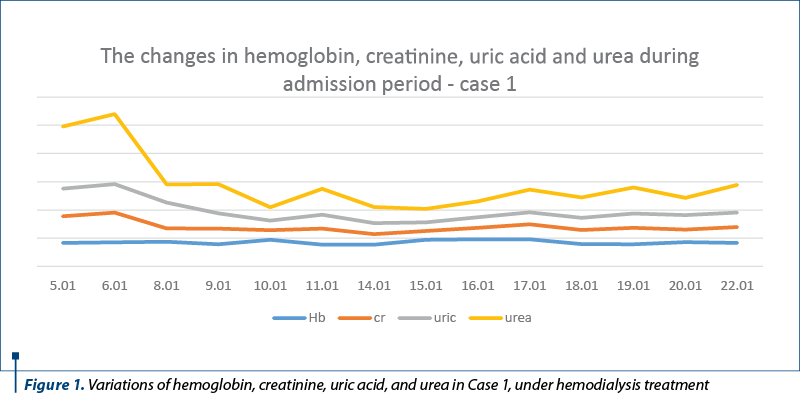
Despite the hemodialysis treatment and her antihypertensive drugs, her blood pressure became impossible to control, interchanging different medication, and the immediate intervention was needed – she delivered by caesarean section at 32 weeks of pregnancy, for fetal distress, a 1150 g male, evaluated at 6 in Apgar score at 1 minute and at 7 and 5 minutes, who required intubation in the intensive care unit.
The postoperative recovery was uncomplicated during the first three days, with standard recovery after delivery by caesarean section, until the fourth day, when the patient presented with high blood pressure, nonresponsive to different types of treatment and a headache with frontal-occipital location for which we performed a cranial CT scan, yet with normal results. In the fifth day after the surgery, the patient was transferred to the Nephrology Department of the Bucharest University Emergency Hospital for further investigations and treatment.
Case 2
The second case is a younger patient, a 25-year-old female, with 22 weeks of pregnancy, with end-stage renal disease and hypertensive disorder, anemia and metabolic acidosis. She was referred to the Nephrology Department of the Bucharest University Emergency Hospital by her gynecologist for nitrogen retention, then being transferred in the Department of Obstetrics and Gynecology of the Bucharest University Emergency Hospital. In regards to personal medical history, it is important to mention that the patient was diagnosed since childhood with anemia for which she was treated with iron supplements, not further investigated, and she was also diagnosed with multiple episodes of urinary tract infections accompanied by lumbar pain, not being accompanied by gross hematuria, fever or shiver.
The first renal ultrasound evaluation was two years prior to the admission in our hospital, when she was diagnosed with unilateral right renal hypoplasia, with parenchymal index of 5 mm and a grade 1 ureterohydronephrosis on the left side. In order to better evaluate the function and anatomy of the kidneys, a renal scintigraphy was performed, revealing only a 20% functioning renal cortical tissue on the right side and 80% on the left side.
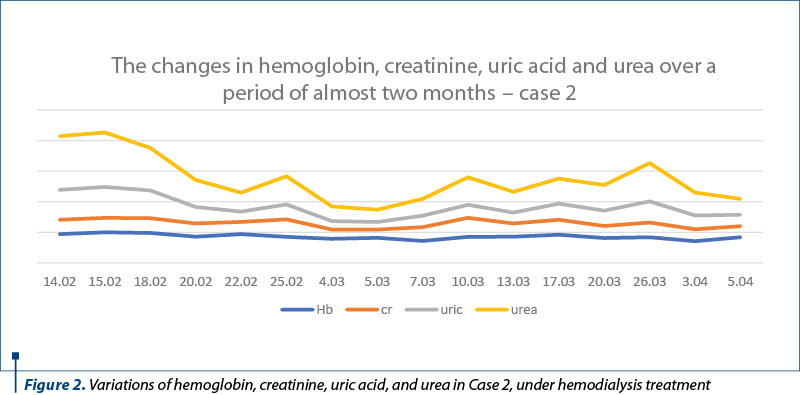
Ever since the first weeks of pregnancy, the patient presented with high blood pressure (max. SBP – 160 mmHg) for which she underwent treatment with methyldopa. Until 18 weeks of pregnancy, the patient did not go to her regular follow-up visits to the gynecologist. The level of hemoglobin was 6.9 g/dl, indicating severe anemia, she had hyperuricemia (10.1 mg/dl), mild hypoalbuminemia (3.4 g/dl), serum protein level was 6.1 g/dl, serum creatinine: 4.56 mg/dl, urea: 149 mg/dl, eGFR: 12 ml/min, and proteinuria: 3736 mg/24 hours. The variations of hemoglobin, creatinine, uric acid, and urea during the admission in the Department of Obstetrics and Gynecology of the Bucharest University Emergency Hospital are presented in Figure 2. During admission in the hospital, it was administered antihypertensive treatment, two units of blood transfusion, recombinant human erythropoietin, and alkali treatment for metabolic acidosis. Several ultrasound examinations of the kidneys have been performed (Figure 3).
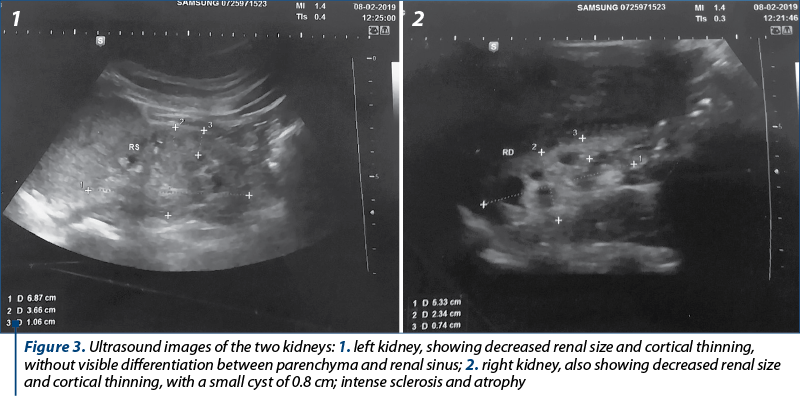
During admission period, she was under hemodialysis treatment once a day or once every two days. She delivered by caesarean section, at 31 weeks of pregnancy for fetal distress, a 1005 g male baby, evaluated at 4 in Apgar score at 1 minute and at 6 and 5 minutes. The postoperative evolution of the mother was favorable. The newborn was admitted in the Neonatal Intensive Care Unit, and he also had a good evolution.
Discussion
CKD is defined as an abnormal structure or function of the kidney, which persists for more than three months, with implications on health condition. The glomerular filtration rate (GFR) indicates the condition of the excretory function and is considered the best overall index of kidney function, at the same time being an easy to measure index, from the results of blood creatinine test, age, body size and gender. In pregnancy, GFR increases by approximately 50%, resulting in a physiologic reduction in serum creatinine. Damage in the kidney structure or function, whether it’s about parenchymal structure, blood vessels or collecting systems, is rarely diagnosed by direct examination of kidney tissue, as it is often suggested by markers of renal function(11,12). Imbasciati et al. reports an increased risk for progression to dialysis in patients with GFR <40 ml/min and proteinuria over 1 g/day(13).
Chronic kidney disease may present with a large variety of causes, the most frequent being diabetes, high blood pressure, polycystic kidney or glomerulonephritis, yet the exact evidence of kidney damage during pregnancy is difficult to obtain because of the limitation on radiologic tests that can be performed during this time(14). In our case, the scintigraphy evaluation for renal functionality was performed prior to the moment of conceiving. The risk for adverse pregnancy outcome increases from stage 1 to stage 5 CKD and may be higher in glomerular nephropathies, autoimmune disease or diabetes(15).
Pre-pregnancy evaluation is needed in order to optimize the end result of the whole process, as the best outcome for the mother and for the fetus is desirable. The lack of clear evidence for the best methods in taking care of such cases, being more difficult as the stage of CKD is more advanced, forces the physician to find more appropriate management methods. Thus, the approach may differ from one center to another, being more suitable as the team is multidisciplinary, including the obstetrician, nephrologist, maternal-fetal medicine specialist, and neonatal intensive care unit(16).
As mentioned before, pre-conceiving evaluation also needs to include proper counseling as patients with CKD need to be informed about the risk involved, regarding worsening of their renal condition, the fetal risks (i.e., prematurity), but also the possible teratogenic effect of the medication, therefore the drugs that may affect the baby should be avoided. Patients who have benefited from these information declared a positive experience(10,17). The term “obstetric nephrology” has been proposed to identify the importance of this binomial state(18).
In the last years, there have been published numerous studies presenting the bidirectional impact regarding the CKD and the pregnancy – by the high risk pregnancy in women with CKD through hypertension and proteinuria, but also by the negative impact of the pregnancy on the already damaged renal function and structure, as these became more frequent by obtaining a pregnancy even in the end stage of the renal disease. As mentioned before, even the first stages of renal disease predispose both the mother and the fetus to increasing morbidity and mortality(19).
It is known that the risk increases with the degree of renal insufficiency and is further heightened by coexisting hypertension and proteinuria(4). As such, pregnancies occurring in young women with advanced renal insufficiency can result in significantly compromised maternal and fetal well-being.
According to Piccoli et al., CKD outcome was worse than physiologic pregnancies: preterm delivery (44% vs. 5%) was more frequent, such as the need for performing caesarean section (44% vs. 25%), and for neonatal intensive care (26% vs. 1%)(20). These differences were significant even for the stage 1 of CKD, being more pronounced as the severity increased. Those results were also correlated with the severity of proteinuria(21). The adverse maternal and fetal events of CKD during pregnancy are presented in Table 1.

The pregnant patient with chronic kidney disease represents a challenging case, because there is insufficient experience in managing such cases, being more difficult as the stage of CKD is more advanced; but nowadays, the incidence of these cases is increasing, mostly due to an effective collaboration between the obstetrician, nephrologist, maternal-fetal medicine specialists and neonatologist, and conceiving even in the end stage renal disease is not an uncommon event(16).
Another study, published in 2015, that analyzed 80 pregnant patients over a period of 11 years, showed similar results on the overall feto-maternal outcome. Regarding the maternal complications, the results indicated an increased risk for preeclampsia, severe anemia (p=0.001), and deterioration of kidney function, more severe in advanced stages of CKD. Fetal short-term outcome was also affected, the complications consisting in intrauterine growth restriction, low Apgar score at 5 minutes, or need for intensive care unit; all the complications were encountered with increased frequency when compared to the general population(22). The prevalence of small-for-gestational-age (SGA) babies has been shown to be higher in mothers receiving peritoneal dialysis compared with those receiving hemodialysis (66% vs. 31%)(23).
A close monitoring and a more intense dialysis regimen lead to a better outcome of the overall feto-maternal state, mimicking a near normal renal function, disposing the uremic toxins. Thus, it would be created a better environment for the fetus and a proper placental blood flow, even in the advanced CKD(6,24). As early as 1980, the incidence of pregnancy while on dialysis was reported to be 0.9%, increasing in time to 7% by 2003. And is not only the incidence of obtaining a pregnancy that increased, but also the overall survival of the babies, rising from a reported percentage of only 20%(25) to more than 50% survival rate, remaining, of course, a rate of neonatal death higher than in the general population(26).
Another report published in 2012 showed similar results regarding the maternal and fetal outcome. Although on a small number of patients, it concluded that 44% of the patients developed preeclampsia, and 42% suffered adverse perinatal outcome, including four cases of perinatal deaths. The authors concluded that preeclampsia and proteinuria could be two independent risk factors for the adverse perinatal outcome(27).
In present time, Kendrick et al. reported a higher rate of infant survival, up to 71%. Half of the patient were at risk for preterm delivery, with 33% increased odds of caesarean section. Kidney disease was also associated with two-fold increased odds of low birth weight (OR: 2.38; 95% CI; 1.64-3.44)(28).
Fink et al. confirmed that women with kidney disease had a higher risk of preterm delivery and of caesarean section compared to women without chronic kidney damage(29). These data demonstrate that women with underlying renal disease are at increased risk for adverse maternal and perinatal outcomes.
It would also be of interest to observe the long-term consequences of the children born from CKD mothers. Even though most of the studies focus on the short-term outcomes for the children and mother, it is also important how the future life of these children will be affected, considering that preterm delivery or IUGR predispose them to cardiovascular and renal disease as adults(2).
Conclusions
Adverse maternal and fetal outcomes remain high in women with chronic kidney disease, as they are more likely to have preterm deliveries with low birth weight infants and deliver by caesarean section compared to women without kidney disease.
CKD is a challenge for pregnancy even from early stages, and strict and individualized monitoring is required. These patients need more resources and frequent hospitalizations, with higher charges than for women without kidney disease.
Conflict of interests: The authors declare no conflict of interests.
Bibliografie
- Lim V, Henriquez C, Sievertsen G, Frohman L. Ovarian function in chronic renal failure: evidence suggesting hypothalamic anovulation. Annals of Internal Medicine. 1980; 93(Part 1):21-7.
- Haesler E, Melhem N, Sinha M. Renal disease in pregnancy: fetal, neonatal and long-term outcomes. Best Practice & Research Clinical Obstetrics & Gynaecology. 2019; Available at: https://www.sciencedirect.com/science/article/pii/S1521693418302086?dgcid=rss_sd_all
- Williams D, Davison J. Chronic kidney disease in pregnancy. BMJ. 2008; 336, 211–215.
- Suarez M, Kattah A, Grande J, Garovic V. Renal Disorders in Pregnancy: Core Curriculum 2019. Am J Kidney Dis. 2019; 73(1):119-30.
- Hladunewich M, Schatell D. Intensive dialysis and pregnancy. Hemodial. Int. 2016; 20:339–48.
- Alkhunaizi A, Melamed N, Hladunewich M. Pregnancy in advanced chronic kidney disease and end-stage renal disease. Current Opinion in Nephrology and Hypertension. 2015; l24(3):252-9.
- Sato J, De Oliveira L, Kirsztajn G, Sass N. Chronic kidney disease in pregnancy requiring first-timedialysis. Int J Gynaecol Obstet. 2010; 111:45–8.
- Bar J, Orvieto R, Shalev Y, Peled Y, Pardo Y, Gafter U, Hod M. Pregnancy outcome in women with primary renal disease. Israel Med Assoc J. 2000; 2(2):178-81.
- Hladunewich M. Chronic kidney disease and pregnancy. In: Seminars in Nephrology. WB Saunders, 2017; 37(4):337-46.
- Wiles K, Bramham K, Vais A, Harding K, Chowdhury P, Taylor CJ, Nelson-Piercy C. Pre-pregnancy counseling for women with chronic kidney disease: a retrospective analysis of nine years experience. BMC Nephrology. 2015; 16(1):28.
- Chapter 1. Definition and classification of CKD. Kidney Int Suppl. 2013; 3(1):19-62.
- Levey A, Coresh J, Balk E, Kausz A, Levin A, Steffes M, Eknoyan G. National Kidney Foundation practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Ann Inter Med. 2003; 139(2):137-47.
- Imbasciati E, Gregorini G, Cabiddu G, Gammaro L, Ambroso G, Del Giudice A, Gravidanza R. Pregnancy in CKD stages 3 to 5: fetal and maternal outcomes. Am J Kidney Dis. 2007; 49(6):753-62.
- Cunningham F, Leveno K, Bloom S, Hauth J, Rouse D, Spong C. Implantation, embryogenesis, and placental development. Williams Obstetrics. 2005; 22:39-90.
- Piccoli G, Alrukhaimi M, Liu Z, Zakharova E, Levin A. What we do and do not know about women and kidney diseases; questions unanswered and answers unquestioned: reflection on World Kidney Day and International Woman’s Day. Nephrology (Carlton). 2018 Mar; 23(3):199-209.
- Hladunewich M, Melamad N, Bramham K. Pregnancy across the spectrum of chronic kidney disease. Kidney Int. 2016; 89(5):995-1007.
- Chinnappa V, Ankichetty S, Angle P, Halpern S. Chronic kidney disease in pregnancy. Int J Obstet Anest. 2013; 22(3):223-30.
- August P. Obstetric nephrology: Pregnancy and the kidney – inextricably linked. Clin J Am Soc Nephrol. 2012; 7:2071–2.
- Koratala A, Bhattacharya D, Kazory A. Chronic Kidney Disease in Pregnancy. South Med J. 2017; 110(9):578-85.
- Piccoli G, Fassio F, Attini R, Parisi S, Biolcati M, Ferraresi M, Todros T. Pregnancy in CKD: whom should we follow and why?. Nephrol Dialysis Transpl. 2012; 27 (suppl 3):iii111-iii118.
- Piccoli G, Attini R, Vasario E, Conijn A, Biolcati M, D’Amico F, Todros T. Pregnancy and chronic kidney disease: a challenge in all CKD stages. Clin J Am Soc Nephrol. 2010; 5(5): 844-55.
- Bharti J, Vatsa R, Singhal S, Roy K, Kumar S, Perumal V, Meena J. Pregnancy with chronic kidney disease: maternal and fetal outcome. Eur J Obstet Gynecol and Reprod Biol. 2016; 204:83-7.
- Piccoli G, Minelli F, Versino E, Cabiddu G, Attini R, Vigotti F, Todros T. Pregnancy in dialysis patients in the new millennium: a systematic review and meta-regression analysis correlating dialysis schedules and pregnancy outcomes. Nephrol Dialysis Transpl. 2015; 31(11):1915-34.
- Inal S, Reis K, Armağan B, Oneç K, Biri A. Successful pregnancy in an end-stage renal disease patient on peritoneal dialysis. In: Advances in Peritoneal Dialysis. Conference on Peritoneal Dialysis. 2012; 28:140-1.
- Wing A, Brunner F, Brynger H, Chantler C, Donckerwoicke R, Gurland H, Mansell M. A. Successful pregnancies in women treated by dialysis and kidney transplantation. Brit J Obstet Gynaecol. 1980; 87:839-45.
- Holley J, Reddy S. Pregnancy in dialysis patients: a review of outcomes, complications, and management. In: Seminars in Dialysis. 2003; 16(5):384-8.
- Xie M, Zhang C, Wang J, Wang S, Zhang X. Analysis of the perinatal outcome and risk factors for pregnancies complicated with chronic renal diseases. Zhonghua Fu Chan Ke Za Zhi. 2012; 47(3):161-5.
- Kendrick J, Sharma S, Holmen J, Palit S, Nuccio E, Chonchol M. Kidney disease and maternal and fetal outcomes in pregnancy. Am J Kidney Dis. 2015; 66(1):55-9.
- Fink J, Schwartz S, Benedetti T, Stehman-Breen C. Increased risk of adverse maternal and infant outcomes among women with renal disease. Paediatr Perinat Epidemiol. 1998; 12(3):277-87.
Articole din ediţiile anterioare
Sarcina ectopică cu evoluţie atipică
Prezentăm cazul unei paciente în vârstă de 32 de ani, cu istoric de infertilitate de cauză neexplicată (2 inseminări şi 2 fertilizări in vitro pe c...
Managementul ante-, intra- şi post-partum al unei adolescente gravide diagnosticate cu limfom Hodgkin
Limfomul Hodgkin este o reală problemă de sănătate, deoarece incidenţa sa atinge apogeul în perioada adolescenţei, în special subtipul clasic cu sc...
Complicaţiile perinatale şi neonatale la pacientele cu răspuns ovarian scăzut în sarcinile obţinute prin proceduri de reproducere umană asistată
In vitro fertilization (IVF) technologies with a controlled ovarian hyperstimulation approach have classified patients into three different groups ...
Monitorizarea funcţiei tiroidiene în sarcină
Patologia glandei tiroide este una dintre cele mai frecvente afecţiuni ale sarcinii, cu repercusiuni deopotrivă materne şi fetale. Hormonii tiroidi...