SĂNĂTATE PUBLICĂ
Tuberculoză cu germeni rezistenți la mai multe medicamente antituberculoase. Evaluare rapidă a riscului.
Multidrug-resistant tuberculosis in migrants, multi-country cluster
Abstract
A multi-country cluster of multidrug-resistant tuberculosis (MDR TB) involving 28 migrants has been delineated by whole genome sequencing (WGS). In December 2016, Switzerland initially reported a cluster of seven MDR TB cases to the European Commission in newly-arrived migrants from Somalia (5 cases), Eritrea (1 case) and Ethiopia (1 case). The Commission then informed the Member States through an Early Warning and Response System (EWRS) message. Following the EWRS notification, Germany, Austria, Finland, France and Sweden reported cases that were linked to this cluster on the basis of WGS. Switzerland later reported an eighth case. As of 4 April 2017, isolates from 28 cases that are part of the WGS cluster had been reported from Germany (14), Switzerland (8), Austria (2), France (2), Finland (1) and Sweden (1). All cases have a recent history of migration from Somalia (23), Eritrea (3), Sudan (1) and Ethiopia (1).The six countries involved in the multi-country cluster have implemented migrant screening. Early case identification of active TB and drug susceptibility testing, especially in migrants arriving from the Horn of Africa, is important in order to identify and treat active cases and provide preventive treatment or monitoring for those diagnosed with latent tuberculosis infection[10]. It is therefore important to rapidly investigate exposure risk factors, including the travel history and itineraries of patients and their contacts, and share this information to determine whether transmission may have taken place in the EU/EEA, during migration, or in the country of origin. Depending on the results of the investigation, appropriate prevention and control measures should be taken. Although the number of cases detected so far suggests that there is only a limited risk of this cluster becoming a widespread event in Europe, more cases associated with this cluster may yet be identified.
Keywords
Rezumat
Tulpinile de la un grup de 28 de migranți cu tuberculoză rezistentă la mai multe medicamente anti-TB (MDR-TB) au fost secvențiate (prin secvențierea întregului genom, WGS).În decembrie 2016, Elveția a raportat la CE/ECDC un grup de șapte cazuri de MDR TB – migranți nou veniți din Somalia (5 cazuri), Eritreea (1 caz) și Etiopia (1 caz). Comisia a informat statele membre printr-un mesaj transmis prin sistemul rapid de informare și răspuns (EWRS; Early Warning and Response System). După notificarea prin EWRS, Germania, Austria, Finlanda, Franța și Suedia au raportat cazuri legate de acest grup în baza datelor oferite prin WGS. Elveția a raportat mai târziu un al optulea caz. Tulpinile izolate de la cele 28 de cazuri sunt distribuite după cum urmează: Germania (14), Elveția (8), Austria (2), Franța (2), Finlanda (1) și Suedia (1). Toate cazurile au o istorie recentă a migrației din Somalia (23), Eritreea (3), Sudan (1) și Etiopia (1). Cele șase țări europene au implementat screening-ul migranților.
Identificarea timpurie a cazurilor de TB activă și testarea sensibilității la chimioterapice anti-TB, în special în cazul migranților care sosesc din Cornul Africii, sunt importante pentru a identifica și trata cazurile active și pentru a oferi tratament preventiv sau monitorizare pentru cei diagnosticați cu tuberculoză latentă[10].
Prin urmare, este important să se investigheze rapid factorii de risc, inclusiv istoricul călătoriilor și itinerariile pacienților și contactele acestora. Totodată este important să se comunice aceste informații pentru a se putea stabili dacă transmiterea a avut loc în Uniunea Europeană/Aria Economică Europeană (EU/EEA), în timpul migrației sau în țara de origine. În funcție de rezultatele investigației, trebuie luate măsuri adecvate de prevenire și control.
Cu toate că numărul de cazuri detectate până acum sugerează că riscul actual este limitat, este posibil ca în următoarea perioadă să fie identificate și alte cazuri asociate cu acest grup.
Cuvinte Cheie
Public health issue
This third update provides information on the risk of transmission within the EU of an MDR TB clone initially detected in seven asylum seekers from the Horn of Africa currently residing in Switzerland. Recommendations are given to help improve the understanding and the public health impact of this cluster for the EU.Consulted experts
ECDC internal response team in alphabetical order: Sergio Brusin, Mike Catchpole, Denis Coulombier, Josep Jansa, Daniel Palm, Marieke van der Werf. Experts and institutions contributing to this risk assessment: Peter Helbling, Federal Office of Public Health, Switzerland, Stefan Kröger, Robert Koch Institute, Germany, Hanna Soini, National Institute for Health and Welfare, Finland, Ramona Groenheit, Public Health Agency of Sweden, Jean-Paul Guthmann, Santé Publique, France, Pr Jérôme Robert, French Mycobacteria National Reference Laboratory and Lena Fiebig, Robert Koch Institute, Germany.Disease background information
Multidrug-resistant tuberculosis (MDR TB) is defined as tuberculosis disease caused by a Mycobacterium tuberculosis complex strain resistant to at least rifampicin and isoniazid[1]. MDR TB is an urgent public health priority in Europe, with significant health and cost implications associated with the expensive and prolonged treatment often required[2]. Inadequate or incomplete TB treatment is the main risk factor for the development of resistance among TB cases and is usually associated with intermittent drug use, errors in medical prescription, poor patient adherence and low quality of TB drugs[3].Options for prevention of TB infection among contacts of MDR TB cases are limited and require an individual risk assessment, taking into consideration:
risk of progression to TB disease;
drug susceptibility pattern of the source case; and
risk of adverse drug events[4,5].
Migrants seeking refuge from conflict or from deprived areas may be at increased risk of TB and MDR TB because of the collapse of health service infrastructure in these contexts. Some migrant groups, including refugees, rejected asylum seekers, victims of trafficking and undocumented migrants, may be at particularly high risk of (MDR) TB due to poor social conditions (e.g. overcrowding, poor living conditions, incarceration or detention, and homelessness), destitution, exposure to other migrants from high-incidence countries affected by MDR TB along their migration route (or after entry into the host country), or co-infection (e.g. with human immunodeficiency virus)[2].
The burden of tuberculosis in high-income countries disproportionally affects the foreign-born migrant population, and transmission is documented to predominantly occur within migrant communities or indigenous communities, and less between migrant and indigenous communities[2,6]. Active disease occurs in five to ten per cent of those infected from a few months to many years after infection and, in up to ten per cent of those who are HIV-positive per year.
According to data published in the latest WHO TB report, the estimated incidence of TB in Somalia was 274 cases per 100 000 population in 2015. According to the same source, MDR TB was estimated to be the cause in 8.7% of new TB cases. MDR TB was also identified in 47% of previously treated TB cases in Somalia.
Event background information
In November 2016, Switzerland initially reported a cluster of seven MDR TB cases to the European Commission in newly-arrived migrants from Somalia (5 cases), Eritrea (1 case) and Ethiopia (1 case). The Commission then informed the Member States through an Early Warning and Response System (EWRS) message. Following the EWRS notification, Germany, Austria, Finland, France and Sweden reported cases that were linked to this cluster on the basis of WGS. Switzerland later reported an eighth case. As of 4 April 2017, isolates from 28 cases that are part of the WGS cluster had been reported from Germany (14), Switzerland (8), Austria (2), France (2), Finland (1) and Sweden (1). All cases have a recent history of migration from Somalia (23), Eritrea (3), Sudan (1) and Ethiopia (1).
Microbiological investigations
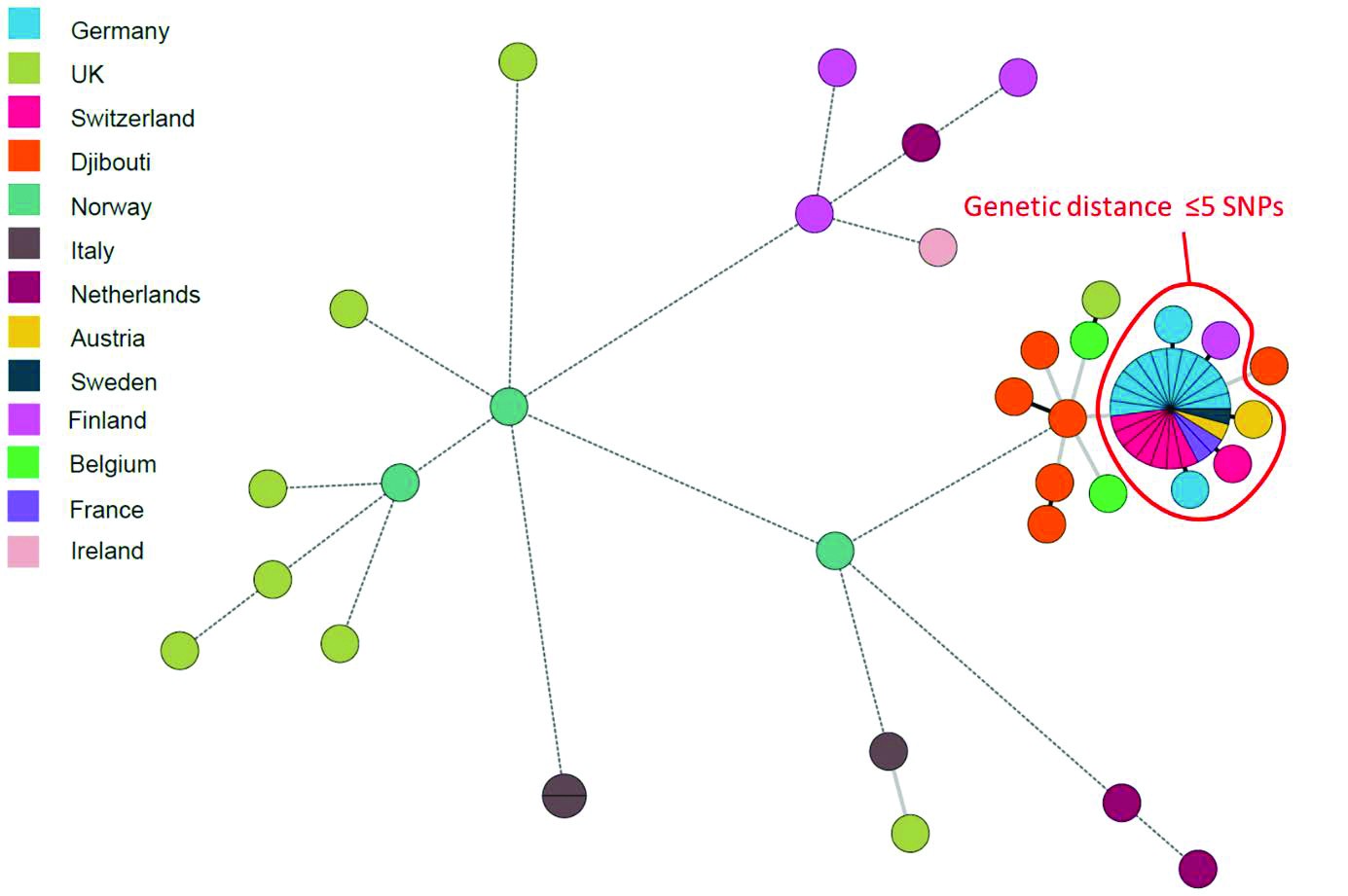
Strains from cases suspected to be part of the cluster were sequenced using the Whole Genome Sequencing (WGS) methodology in the five countries reporting suspected cases. The results were made available to the German National Reference Laboratory for Mycobacteria (Forschungszentrum Borstel, Germany) for analysis. WGS results of strains with similar drug resistance or MIRU-VNTR patterns of cases not linked to the cluster were also included in the analysis to identify potential sources or relatedness (from Belgium, Ireland, Italy, the Netherlands, Norway and the UK). In addition, WGS results of six strains from Djibouti were included in the analysis.Typing data from 2 828 MDR TB isolates reported to ECDC, covering the period 2003 to 2015, show that the cluster strain is rare. Only two MDR TB isolates with the same MIRU-VNTR 24 loci pattern have been reported, both from Belgium. In the first case, diagnosed in 2011, the country of origin was Somalia and in the second, diagnosed in 2013, the country of origin was Djibouti. According to the European Reference Laboratory Network for Tuberculosis, a difference in fewer than six single-nucleotide polymorphisms (SNPs) among isolates may indicate recent transmission[7].
The German National Reference Laboratory for Mycobacteria used an SNP pipeline covering 1 157 potential variable positions in the Mycobacterium tuberculosis genome in their WGS analysis.
The minimum spanning tree in Figure 1 shows the relatedness of 57 Mycobacterium tuberculosis Cameroon genotype (lineage 4.6.2) clinical isolates from migrants with MDR TB in 2016 and 2017. Phylogenetic inference is based on 1 150 SNPs using BWA (Burrows-Wheeler Aligner) and GATK (Genome Analysis Toolkit) for a reference mapping approach. Solid black branches indicate a genetic distance ≤ 5 SNPs, solid grey branches ≤ 12 SNPs, and dashed grey branches > 12 SNPs. The isolates in the current cluster are separated by only one SNP, indicating a common source of infection within the past two to three years.
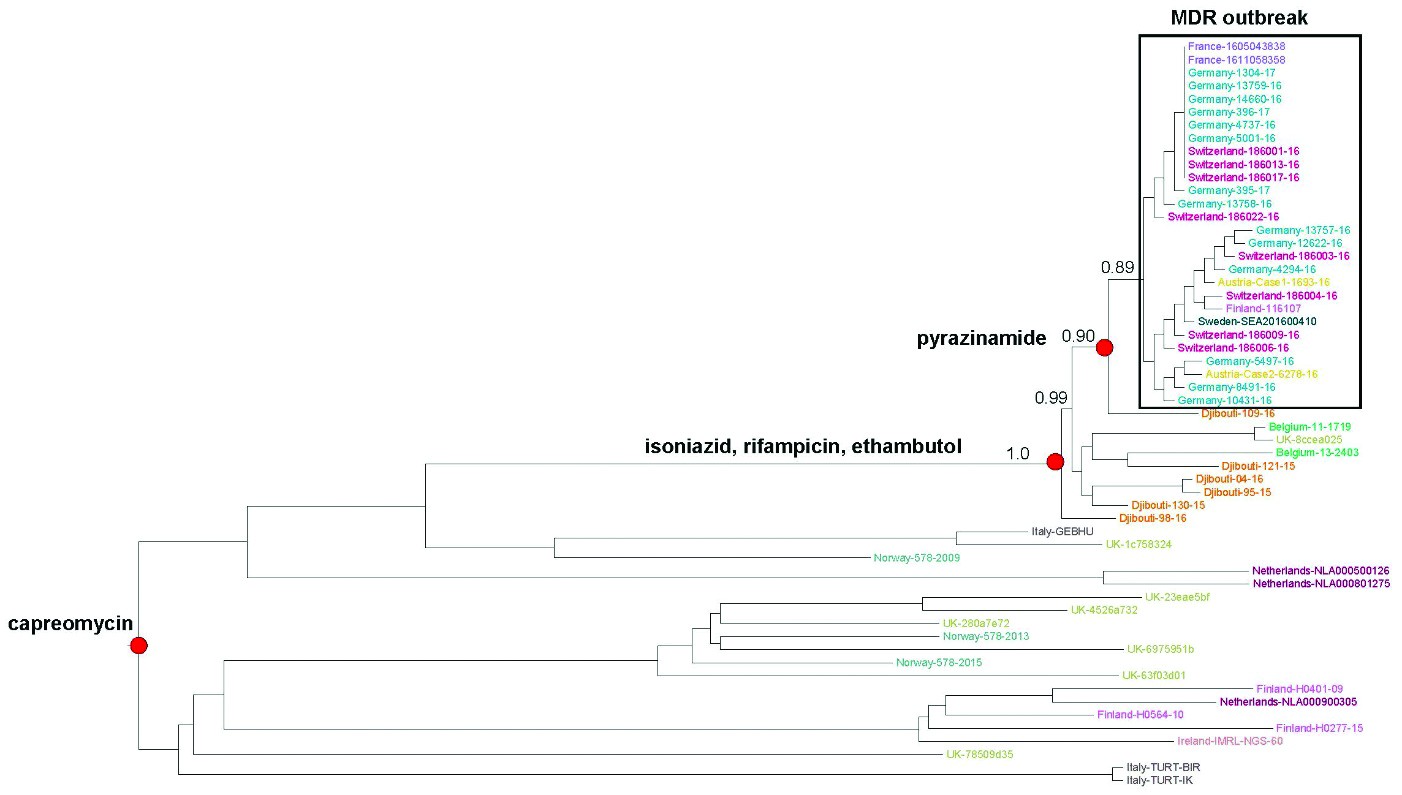
The maximum likelihood phylogeny in Figure 2 was calculated with ‘FastTree’ using the same sequence alignment as in Figure 1. Branches representing the hypothetical progenitors of the identified MDR TB cluster are labelled with confidence values based on 1 000 resamples. In the resulting tree identical isolates are artificially separated by the algorithm, but the confidence of those splits is close to zero and thus not relevant. Resistance acquisition to individual first and second-line anti-TB drugs mediated by distinct mutations is indicated on branches. The maximum likelihood tree shows that the isolate Djibouti 109-16 and the cluster isolates share an immediate common ancestor. Available data suggest that the origin of the cluster strain stems from the Horn of Africa. Further comparisons of WGS profiles from the cluster cases and additional circulating strains in the Horn of Africa could confirm the ancestral relationship and make it possible to find a probable date of divergence for the cluster strain. Available information suggests that divergence from a common ancestral strain took place more than three years ago. Representative sampling and sequencing of isolates from the Horn of Africa that could support this analysis is currently not available, making such an assessment impossible.
Circumstantial epidemiological evidence also supports the hypothesis of an origin of cluster strain in Somalia (see ‘Analysis of migration routes of the cases’). Most of the cases in Germany (13) are recent Somali immigrants and the Somali represent less than 2% of the recent migration flux into Germany (Eurostat: http://ec.europa.eu/eurostat/statistics-explained/index.php/Asylum_statistics). The fact that this cluster has not been observed in migrants of any other nationality suggests a source in Somalia.
Analysis of migration routes of the cases
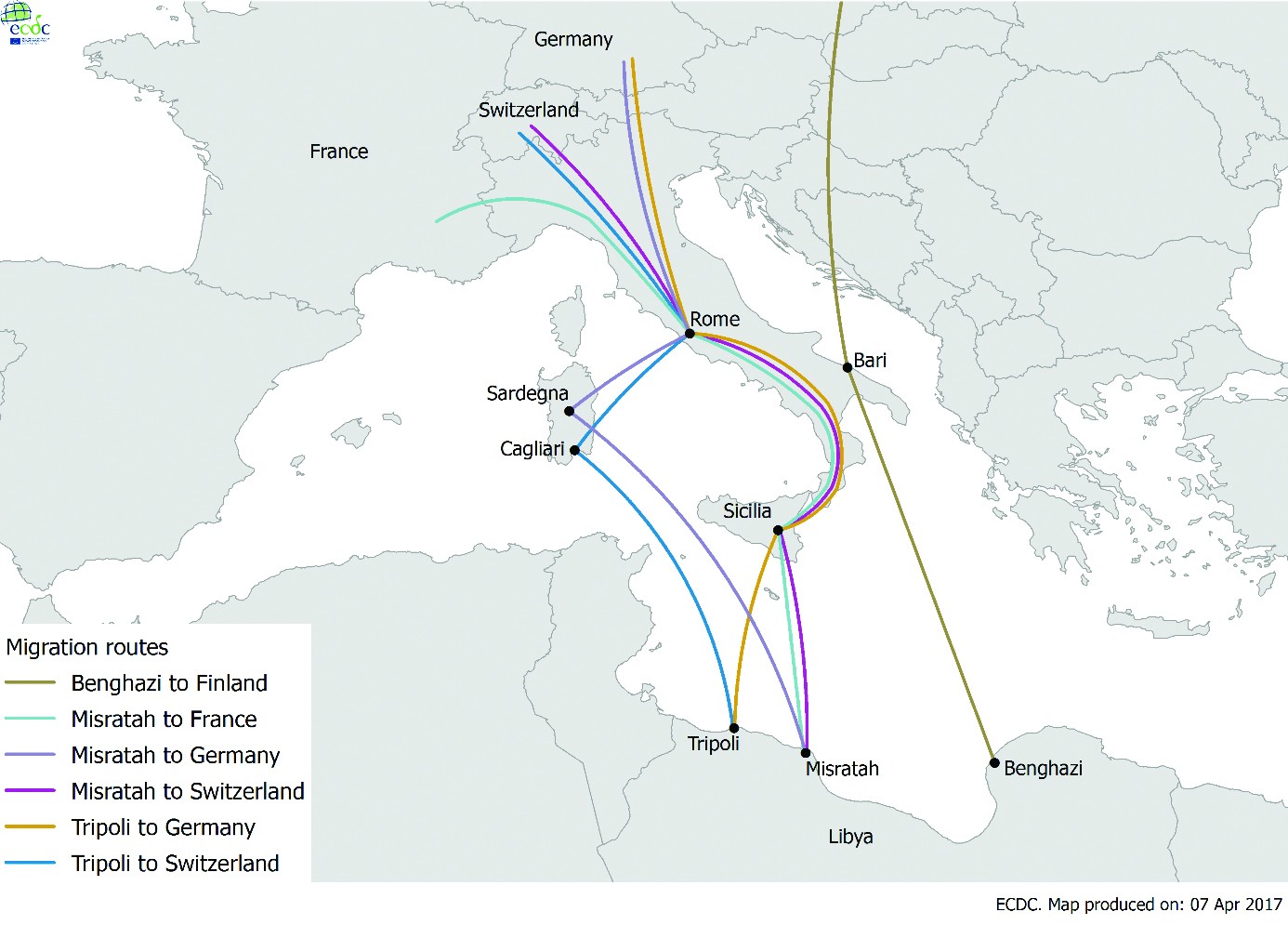
For eight of the migrants, the migration route could not be documented. An analysis of the 20 cases interviewed with documented route of arrival to the EU shows that six cases reported onset of symptoms prior to arrival in the EU, two of which had onset of symptoms in Somalia and one in Ethiopia prior to migration. Five cases reported onset of symptoms during migration, three in Libya and two in Sicily. For the other 12 cases only the date of diagnosis in Europe is known. The two cases that had been symptomatic before leaving Somalia were probably infected while in Somalia and the one that had been symptomatic in Ethiopia was probably infected in Ethiopia.Nineteen of the cases had travelled through Libya between August 2014 and April 2016 and nine of them had had long stays in Bani Walid. The remaining case that reported onset of symptoms before migrating was reported to have travelled between Somalia and Sweden by aeroplane within four days. Three cases reported onset of symptoms while in Libya, with one indicating a stay in Bani Walid.
Eighteen of the cases entered the EU through Italy, one through Germany and one travelled directly to Sweden. In Italy at least six migration routes were mentioned: from Sicily to Switzerland and France, from Sicily to Germany, from Sardinia to Switzerland, from Sardinia to Germany and from Bari directly to Finland (Figure 3). However, all cases but one were in transit through Rome.
ECDC threat assessment for the EU
Twenty-three of the twenty-eight cases in this cluster have an epidemiological link with Somalia, although Somalis represent only 2.1% of the refugee population in the EU/EEA according to the International Organization for Migration (IOM). The three remaining cluster cases had travelled along the same migration route. No cases associated with other migration routes to the EU have been identified in relation to this cluster, suggesting that transmission took place in the country of origin or along the migration route prior to entering the EU/EEA.The clustering of case strains by WGS within one SNP difference suggests that transmission was recent and likely to have taken place either in the patients’ country of origin or in a place along their migration route to the country of destination. For three of the 20 documented cases, infection clearly occurred while in the Horn of Africa, prior to migration. However, in other cases, infection may have occurred during migration, particularly in Libya for migrants who spent a long time there in unsanitary conditions (e.g. Bani Walid). It is possible that some transmissions occurred in Europe, in particular among persons having spent a substantial amount of time in unfavourable conditions at some EU refugee camps. For example, this may have been the case for the Sudanese refugee reported by France, who stayed for one month at a refugee camp in Calais, in the north of France. However, the likelihood of transmission in Europe is considered low, given the efforts to identify cases and the better living conditions in general for migrants in Europe, compared to the conditions during migration.
Infected persons who do not have active TB are not infectious. However, they are at risk of developing active TB disease and becoming infectious. The lifetime risk of reactivation TB for a person with documented latent TB infection is estimated to be 5 to 10%, with the majority developing TB disease within the first five years of initial infection[8].
The risk of transmission within the EU/EEA is primarily within the affected migrant population, but a low risk of transmission to the indigenous population cannot be ignored. This risk is low because TB incidence in a foreign-born population does not have a significant influence on the overall TB incidence in the indigenous population. Therefore, while there remains a risk of additional cases being detected among newly-arrived migrants, the risk of transmission to the EU/EEA population is considered low. n
Mulțumiri: Adresăm mulțumiri Agenției Europene de Prevenire și Control (ECDC) pentru materialele publicate, puse la dispoziția tuturor colegilor din domeniul medical.
Bibliografie
Matteelli A, Roggi A, Carvalho AC. Extensively drug-resistant tuberculosis: epidemiology and management. Clin Epidemiol. 2014;6:111-8.
Hargreaves S, Lonnroth K, Nellums LB, Olaru ID, Nathavitharana RR, Norredam M, et al. Multidrug-resistant tuberculosis and migration to Europe. Clin Microbiol Infect. 2016 Sep 23.
Abubakar I, Zignol M, Falzon D, Raviglione M, Ditiu L, Masham S, et al. Drug-resistant tuberculosis: time for visionary political leadership. Lancet Infect Dis. 2013 Jun;13(6):529-39.
World Health Organization. Guidelines on the management of latent tuberculosis infection. Geneva: WHO; 2015.
European Centre for Disease Prevention and Control. Management of contacts of MDR TB and XDR TB patients. Stockholm: ECDC; 2012.
Pareek M, Greenaway C, Noori T, Munoz J, Zenner D. The impact of migration on tuberculosis epidemiology and control in high-income countries: a review. BMC Med. 2016 Mar 23;14:48.
Nikolayevskyy V, Kranzer K, Niemann S, Drobniewski F. Whole genome sequencing of Mycobacterium tuberculosis for detection of recent transmission and tracing outbreaks: A systematic review. Tuberculosis (Edinburgh, Scotland). 2016 May;98:77-85.
Comstock GW, Livesay VT, Woolpert SF. The prognosis of a positive tuberculin reaction in childhood and adolescence. Am J Epidemiol. 1974 Feb;99(2):131-8.
World Health Organization. Systematic screening for active tuberculosis. Principles and recommendations. Geneva: WHO; 2013.
Lonnroth K, Migliori GB, Abubakar I, D'Ambrosio L, de Vries G, Diel R, et al. Towards tuberculosis elimination: an action framework for low-incidence countries. Eur Respir J. 2015 Apr;45(4):928-52.
Articole din ediția curentă
PREZENTARE DE CAZ
Microcalorimetria – o metodă cu potenţial în diagnosticul infecţiilor ortopedice
Mihnea Gabriel Popa, Zsombor Panti, Mihai Nica, Marian Pleniceanu, Anda Băicuș, Vlad Tudor Popa, Răzvan Ene, Cătălin Cîrstoiu
Microcalorimetry – a promising method for the diagnosis of orthopedic infections Suggested citation for this article: Popa MG, Pa...
STUDII ORIGINALE
Date cu privire la impactul diagnostic generat de prezența anticorpilor anti CMV IgM în sarcină
Loredana-Sabina-Cornelia Manolescu, Anda Dragodan, Gabriela-Loredana Popa
Date cu privire la impactul diagnostic generat de prezența anticorpilor anti CMV IgM în sarcină Suggested citation for this article: Manolescu LSC, Dragodana A, Popa GL. The diagnostic impact generat...
STUDII ORIGINALE
Prevalenţa infecţiei cu virusuri hepatitice la pacienţii cu lichen plan
Mădălina Irina Mitran, Ilinca Nicolae, Mircea Tampa, Cristina Iulia Mitran, Mircea Ioan Popa, Simona Roxana Georgescu
The prevalence of infection with hepatitis viruses in patients with lichen planus Suggested citation for this article: Mitran MI, Nicolae I, Tampa M. The prevalence of infection with hepatitis viruse...Articole din edițiile anterioare