Carcinom spinocelular dezvoltat pe leziuni de lichen plan oral – prezentare de caz şi revizuirea literaturii de specialitate
Squamous cell carcinoma arising in oral lichen planus – case report and literature review
Abstract
Background. Oral lichen planus (OLP) is a relatively common chronic inflammatory disease that usually affects middle-aged individuals and typically follows a prolonged course. It carries a higher risk of complications than its cutaneous counterpart, the most important being malignant transformation. Case report. We present the case of a 64-year-old female patient who developed oral squamous cell carcinoma at the site of a preexisting OLP lesion, and we discuss the risk factors and the mechanisms believed to be responsible for the malignant transformation of OLP, the clues for the early detection of oral cancer and the adequate management of these patients. Conclusions. The difficulty of establishing the diagnosis, the lack of preventive measures and the curative treatment for OLP make regular follow-up essential for these patients. The predisposing factors and the pathways involved in the malignant transformation of OLP lesions need to be further studied.Keywords
oral lichen planusoral cancersquamous cell carcinomaoral lichenoid lesionsleukoplakiaRezumat
Introducere. Lichenul plan oral (OLP) reprezintă o afecţiune inflamatorie cronică relativ frecventă, ce afectează în general indivizi de vârstă medie şi prezintă o perioadă îndelungată de evoluţie. OLP se asociază cu un risc crescut de complicaţii comparativ cu lichenul plan cutanat, cea mai importantă fiind degenerarea malignă. Prezentare de caz. Prezentăm cazul unei paciente în vârstă de 64 de ani, care a dezvoltat carcinom spinocelular la nivelul leziunilor preexistente de OLP şi discutăm factorii de risc şi mecanismele responsabile pentru transformarea malignă a OLP, indiciile utile în detectarea precoce a cancerului oral şi managementul optim al acestor cazuri. Concluzii. Date fiind dificultatea stabilirii diagnosticului şi absenţa unor măsuri de prevenţie şi a unui tratament curativ pentru OLP, monitorizarea atentă a acestor pacienţi este esenţială. Sunt necesare studii suplimentare pentru clarificarea factorilor predispozanţi şi a căilor patogenice implicate în transformarea malignă a leziunilor de OLP.Cuvinte Cheie
lichen plan oralcancer oralcarcinom spinocelularleziuni orale lichenoideleucoplazieIntroduction
Oral lichen planus (OLP) is a relatively common chronic inflammatory disease that involves the oral mucous membranes. Its estimated prevalence varies between 0.1% and 4%(1). OLP usually affects middle-aged individuals, being extremely rare in children, and has a predilection for the female gender, with a female/male ratio of 2:1(2,3). Its etiopathogenesis is not yet completely elucidated.
It represents a multifactorial process, a complex interplay between genetic susceptibility factors and potential environmental triggers, such as infections – especially hepatitis C virus (HCV), herpesviridae and human papillomavirus (HPV) –, dental materials (dental amalgam, metals, resins, composite restorations), systemic medication (antimalarials, gold salts, nonsteroidal anti-inflammatory drugs, beta blockers, diuretics, angiotensin-converting enzyme inhibitors, oral hypoglycemics, penicillamine, oral retroviral medications), psychological stress, trauma, and food allergies that lead to an abnormal T cell immune response(4-7).
The oral cavity has numerous anatomic particularities and various physiologic functions, therefore it requires special attention in terms of diagnosis and treatment. The clinical picture of OLP is diverse, encompassing six forms, namely the reticular, atrophic, papular, bullous, plaque, and the erosive/ulcerative subtypes(4,8). The lesions are generally bilateral and symmetric and often involve multiple sites. They may be asymptomatic or accompanied by pain or a burning sensation aggravated by local trauma and hot, spicy or acidic foods(4,6). All regions of the oral mucosa can be involved, but the most common sites for OLP lesions are the buccal mucosa, the dorsum of the tongue and the gingiva(9). Approximately 15% of OLP patients also present cutaneous lichen planus lesions, while genital lesions are found in almost 20% of cases(8,10,11). The diagnosis is generally based on the characteristic clinical findings, the histopathological examination being necessary only in atypical cases and to exclude malignancy.
In contrast to the self-limited nature of cutaneous lichen planus, OLP typically follows a prolonged, waxing and waning course, with periods of exacerbations and temporary clinical ameliorations(12). Moreover, OLP has a higher rate of complications than its cutaneous counterpart, the most important being malignant transformation.
The incidence of squamous cell carcinoma (SCC) in OLP patients has been investigated in several studies, but they generated conflicting results. The reported malignization rate of OLP lesions ranges between 0.5% and 12.5%(8,13-16), this great variation being mainly attributable to a lack of uniform clinical and histological criteria for the diagnosis of SCC arising in OLP lesions. The clinical, as well as the histological distinction between OLP, oral lichenoid lesions (OLL) and leukoplakia is often challenging and sometimes impossible. Patients suffering from erythematous or erosive OLP and those with OLP lesions located on the lateral border of the tongue carry the highest risk of oral SCC(11,17). Most tumors that arise in OLP lesions are well-differentiated SCCs(3).
We present the case of a 64-year-old female patient who developed SCC in a preexisting OLP lesion and we discuss the risk factors and the mechanisms believed to be responsible for the malignant transformation of OLP, the clues for the early detection of oral SCC and the adequate management of these patients.
Case report
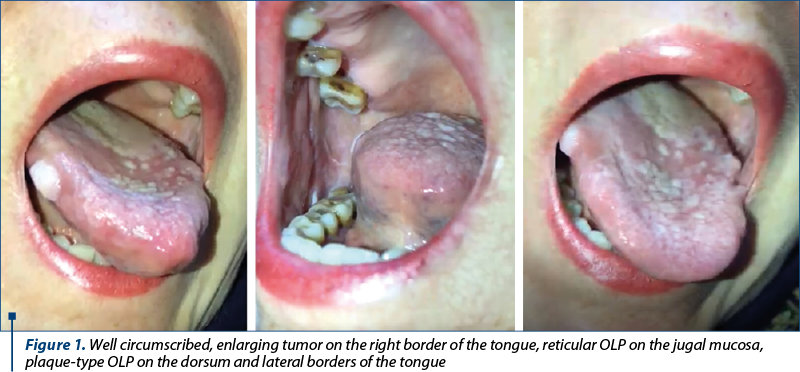
A 64-year-old non-smoking Caucasian female patient was referred to our clinic for the presence of a rapidly enlarging, well circumscribed, oval tumor located on the right lateral border of the tongue, with a diameter of approximately 1/0.7 cm that bled intermittently. Upon physical examination, we noted the presence of interlaced white striae disposed on the jugal mucosa bilaterally and numerous white, homogenous, keratotic plaques of various sizes, ranging from 0.2 to 1 cm, with a tendency to coalesce, located on the dorsal surface and lateral borders of the tongue and on the buccal mucosa (Figure 1). The patient complained of intense pain and burning sensation in the oral mucosa, as well as stiffness that impaired opening of the mouth and made alimentation and oral hygene difficult. The rest of the physical examination was within normal limits, no cutaneous lesions or involvement of other mucosal sites being identified.
The patient had been diagnosed with cutaneous and oral lichen planus three years previously, when she presented to the dermatologist for a cutaneous eruption composed of plane, purple, pruritic papules located around the ankles, on the volar surface of the wrists, and in the lumbosacral region, as well as asymptomatic white striae on the buccal mucosa. The diagnosis of OLP was confirmed by histopathological examination that revealed minimal parakeratosis, acanthosis, a minimally represented granular layer, vacuolar degenerescence of the basal layer, rare apoptotic keratinocytes, lamina propria with slightly densified collagen in the superficial region, as well as a marked, band-like lymphocytic infiltrate. The patient was prescribed local corticosteroid treatment, which led to the remission of the cutaneous lesions and the temporary improvement of the ones involving the oral cavity. During the past three years, the patient intermittently used topical steroids for flares of OLP, but did not present new cutaneous lichen planus lesions. In the previous several months, the oral lesions progressively increased in number and size and became symptomatic and the tumor described above developed.
The patient’s relevant medical history also included arterial hypertension, type 2 diabetes and hypercholesterolemia, for which she was undergoing treatment with oral hypoglicemics, antihypertensive drugs and hypolipemiants.
The results of the laboratory analyses were within normal limits, except for the slightly increased serum level of cholesterol and tryglicerides and mild hyperglicemia. The levels of tumoral markers such as carcinoembryonic antigen and squamous cell carcinoma antigen were also in the normal range. The infection with hepatitis C virus was excluded.
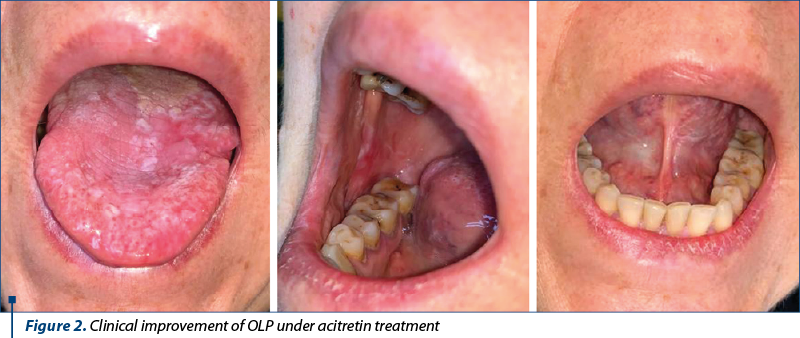
The treatment with oral acitretin (20 mg/day) was initiated and the patient was urgently referred to the oral surgery department. Consecutively, an en bloc partial glossectomy that incorporated the entire tumor located on the right border of the tongue was performed. The histopathological examination showed parakeratosis, papillomatosis, acanthosis with enlarged ridges extending in the dermis (“pushing effect”), and a proliferation of dysplastic, slightly enlarged keratinocytes with abundant cytoplasm, involving the whole thickness of the epidermis, the presence of keratin pearls, and an intra- and peritumoral inflammatory infiltrate. The resection margins were clear. The histopathologic diagnosis was that of OLP with focal well-differentiated cutaneous squamous cell carcinoma (G1), infiltrative to the striated muscle fibres.
Immunohistochemistry stains were positive for cytokeratin 34 beta E12 in lesional squamous cells, nuclear P53 in basaloid tumor cells, and for Ki-67 in 20-30% of tumor cells, mostly in the basaloid cells, but negative for HPV. The maximal tumor thickness was 3 mm, and the depth of invasion (DOI) was 2 mm, with no lymphovascular or perineural invasion.
The patient was referred to an oncologist and a radiation therapist, who recommended a full body computed tomography scan and bone scintigraphy. The results of both investigations were normal. The patient was diagnosed with oral SCC stage I according to American Joint Committee on Cancer (tumor size ≤2 cm, with DOI ≤5 mm)(18).
Oral acitretin 10-20 mg/day was continued and well tolerated. The patient has been monitored every three months in the dermatology and oral surgery departments. The oral lesions decreased in number and size and did not show any worrying changes so far (Figure 2).
Discussion
In 1910, François Henri Hallopeau reported the first case of gingival cancer, the malignant degeneration occurring in a patient suffering from OLP(19). However, it was only in 1978 that the World Health Organization (WHO) classified OLP as a precancerous lesion. Experts have afterwards decided upon a new terminology, differentiating oral potentially malignant lesions from potentially malignant conditions(20). The former are defined as morphologically altered oral mucosa areas that are at increased risk for malignant transformation compared to their normal counterpart and include leukoplakia and erythroplakia. On the other hand, potentially malignant conditions, like oral submucous fibrosis (OSMF) and OLP, represent generalized states associated with a higher malignancy risk. In these cases, the neoplasm may arise in preexisting lesions or anywhere else in the oral mucosa(21). The risk of oral cancer is also significantly increased in the setting of rare, inherited syndromes, particularly xeroderma pigmentosum and Fanconi’s anemia, chronic immunosuppression, and chronic graft versus host disease(22). Worldwide, more than 250,000 oral cancers and over 125,000 deaths attributed to oral cancer occur each year(23,24). Approximately 80% of oral cancers appear in patients with preexisting potentially malignant lesions or conditions(25). Sadly, despite the advances in diagnostic procedures and therapeutic strategies in the past few decades, the five-year survival rate of these patients remains alarmingly low(26). Therefore, the early recognition of malignant change is crucial for a favorable outcome, the patients with stage I oral cavity cancer having a 70% five-year survival rate, according to the National Cancer Database(27).
The potential for malignant transformation of OLP is still a matter of debate. Eisen et al. monitored 723 patients with OLP for different intervals, ranging from six months to eight years, and found a 0.8% rate of malignant transformation(28). A total of 719 OLP cases were analyzed by Lanfranchi-Tizeira et al.(29) They reported 32 cases of oral cancer arising in OLP lesions (6.51%), all of which developed in plaque, erosive or atrophic forms of OLP. An Italian team studied the risk of oral SCC in 402 patients diagnosed with OLP who were monitored for 2-12 years(13). Nine patients developed oral SCC during the follow-up period, the rate of malignant transformation being higher among women (2.8% versus 1.3% in male patients). They also observed that patients with HCV infection had a three times higher risk of SCC than the non-infected OLP patients. Xue et al. studied 674 patients diagnosed with OLP between 1963 and 2003 and reported an oral cancer rate of 0.65%, SCC mainly arising in erosive or atrophic lesions(30). Noteworthy, one patient presented malignant transformation 21 years after the initial diagnosis of OLP. A prospective study carried out in The Netherlands in 2006, that included 192 patients with OLP and OLL who were monitored for 53 months, reported four malignant transformations of OLL lesions and none in the OLP group(31). Unfortunately, many studies lack sufficient data regarding the clinical and histopathological criteria used for the diagnosis of OLP(32). In most studies, the diagnosis was based solely on clinical examination. Given the significant clinical and histopathological similarity between OLP and other conditions, such as OLL, leukoplakia and lupus erythematosus, the differentiation between these disorders is often very challenging. This is why many authors believe a large proportion of supposed OLP that suffered malignant transformation were, in fact, misdiagnosed and most probably represented lichenoid dysplasia(16,33,34).
The mechanism of malignant transformation in OLP lesions is only partially understood. In some experts’ view, OLP represents a model of inflammation-induced neoplasia(35). The chronic stimulation exerted by inflammatory and stromal cells on epithelial cells alters their growth control, proliferation and differentiation and, together with the effects of oxidative stress, induces DNA damage and malignant transformation(36-39). Other authors consider malignant transformation of OLP lesions as the result of an altered expression of p53 protein and other apoptosis-regulating proteins(40-42). Unlike other potentially malignant disorders(43,44), neither the development of OLP, nor its risk of malignant transformation seem to be related to alcohol consumption, tobacco chewing, or tobacco smoking(2,13,45). HPV infection, especially with HPV-16 and HPV-18 genotypes, is considered a risk factor for the development of oropharyngeal cancers(46,47). HPV DNA has been detected in a high proportion of OLP lesions, especially in atrophic lesions(48). Furthermore, HPV DNA, predominately HPV-16, was identified considerably more often in OLP lesions with dysplasia than in OLP lesions that showed no signs of dysplasia(48,49). Our patient did not present any of the aforementioned risk factors associated with an increased risk of oral cancer.
No clinical aspects, molecular markers or histological features are reliable predictors of malignant transformation in OLP. Hence, no efficient prevention strategies are applicable for OLP patients. No curative treatment exists either. The treatment is not usually recommended in mild, asymptomatic forms of OLP. High-potency topical corticosteroids are the first line of treatment for symptomatic lesions. If satisfactory clinical improvement cannot be achieved using topical corticosteroids, or the patient presents with severe or diffuse erosive oral lesions or multiple sites of the oral cavity are involved, oral retinoids, systemic corticosteroids and immunosuppressive agents, mainly cyclosporin, are indicated(50,51). Other treatment modalities, such as photochemotherapy or laser therapy (308-nm Excimer laser radiation, diode laser, or CO2 laser evaporation), might prove beneficial in severe, refractory cases(50). None of these treatments, however, ensures a long-lasting amelioration of the disease(51). In addition, drug-induced immunosuppression and exposure to ultraviolet radiation may increase the risk of oral cancer(16).
Patients diagnosed with OLP should be monitored at least at 6 to 12 months intervals(52,53). The experts agree that this time period is adequate to assess the early signs of malignant transformation. If the patient becomes symptomatic or notices signs like increase in lesion size or thickness, induration, ulceration or bleeding at the site of OLP lesions or enlarged lymph nodes, the disease is highly active, or the patient is a high-risk individual, more frequent follow-up visits should be planned. Careful visual and tactile examination should be performed. Vital staining with toluidine blue and oral cytology, spectroscopy, fluorescence imaging, optical coherence tomography and other imaging techniques may increase the chance of the early detection of oral cancer(54). A lesion located in the oral cavity that persists for more than three weeks and is red/red and white, ulcerated, lump/indurated should raise a high suspicion of SCC and should be biopsied(55). The regional lymph nodes should also be thoroughly examined as nodal metastasis may occur even in stage I OLP-related oral cancer(56). Therefore, the importance of establishing an early diagnosis for the reduction of morbidity and mortality from oral cancer cannot be emphasized enough.
Conclusions
Despite the continuing debate regarding the actual potential and mechanisms of malignant transformation of OLP lesions, there is sufficient evidence to consider this risk real and to closely monitor patients diagnosed with OLP. The difficulty of establishing the diagnosis, the lack of preventive measures and curative treatment make regular follow-up essential. The predisposing factors and the pathways involved in the malignant transformation of OLP lesions need to be studied further. Research should also focus on the identification of the predictive markers of malignant transformation and on new treatment strategies in order to ensure both the control of symptoms and the prevention of malignant transformation.
Bibliografie
-
Sugerman PB, Savage NW, Walsh LJ, Zhao ZZ, Zhou XJ, Khan A. The pathogenesis of oral lichen planus. Crit Rev Oral Biol Med. 2002; 13(4), 350–65.
-
Shen ZY, Liu W, Zhu LK, Feng JQ, Tang GY, Zhou ZT. A retrospective clinicopathological study on oral lichen planus and malignant transformation: Analysis of 518 cases. Med Oral Patol Oral Cir Bucal. 2012; 17(6), e943–e947.
-
Alrashdan MS, Cirillo N, McCullough M. Oral lichen planus: a literature review and update. Archives of Dermatological Research. 2016; 308(8), 539-51.
-
Krupaa RJ, Sankari SL, Masthan KMK, Rajesh E. Oral lichen planus: An overview.
-
J Pharm Bioallied Sci. 2015; 7(Suppl 1), S158–S161.
-
Radochova V, Drizhal I, Slezak R. A retrospective study of 171 patients with oral lichen planus in the East Bohemia - Czech Republic – single center experience.
-
J Clin Exp Dent. 2014; 6(5), e556–e561.
-
Yardimci G, Kutlubav Z, Engin B, Tuzun Y. Precancerous lesions of oral mucosa. World J Clin Cases. 2014; 2(12), 866–72.
-
Serrano-Sánchez P, Bagán JV, Jiménez-Soriano, Sarrión G. Drug-induced oral lichenoid reactions. A literature review. J Clin Exp Dent. 2010; 2(2), e71–e5.
-
Farhi D, Dupin N. Pathophysiology, etiologic factors, and clinical management of oral lichen planus, part I: facts and controversies. Clin Dermatol. 2010; 28(1), 100–108.
-
Oliveira Alves MG, Almeida JD, Balducci I, Guimarães Cabral LA. Oral lichen planus: A retrospective study of 110 Brazilian patients. BMC Res Notes. 2010; 3, 157.
-
Eisen D. The evaluation of cutaneous, genital, scalp, nail, esophageal, and ocular involvement in patients with oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999; 88(4), 431–6.
-
Edwards PC, Kelsch R. Oral lichen planus: Clinical presentation and management.
-
J Can Dent Assoc. 2002; 68(8), 494–9.
-
Eisen D, Carrozzo M, Bagan Sebastian J-V, Thongprasom K. Number V Oral lichen planus: clinical features and management. Oral Dis. 2005; 11(6), 338–49.
-
Gandolfo S, Richiardi L, Carrozzo M, Broccoletti R, Carbone M, Pagano C, Vestita C, Rosso S, Merletti F. Risk of oral squamous cell carcinoma in 402 patients with oral lichen planus: A follow-up study in an Italian population. Oral Oncol. 2004; 40(1), 77–83.
-
Torrente-Castells E, Figueiredo R, Berini-Aytés L, Gay-Escoda C. Clinical features of oral lichen planus. A retrospective study of 65 cases. Med Oral Patol Oral Cir Bucal. 2010; 15(5), e685–690.
-
Lodi G, Scully C, Carrozzo M, Griffiths M, Sugerman PB, Thongprasom K. Current controversies in oral lichen planus: Report of an international consensus meeting. Part 1. Viral infections and etiopathogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005; 100(1), 40–51.
-
Lodi G, Scully C, Carrozzo M, Griffiths M, Sugerman PB, Thongprasom K. Current controversies in oral lichen planus: Report of an international consensus meeting. Part 2. Clinical management and malignant transformation. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005; 100(2), 164-78.
-
Hietanen J, Paasonen MR, Kuhlefelt M, Malmstrom M. A retrospective study of oral lichen planus patients with concurrent or subsequent development of malignancy. Oral Oncol. 1999; 35(3), 278–82.
-
Amin MB, Greene FL, Edge SB, Edge SB, Compton CC, Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR, Winchester DP. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA: Cancer J Clin. 2017; 67(2), 93–9.
-
Hallopeau H. Sur un cas de lichen de Wilson gingival avec néoplasie voisine dans la région maxillaire. Bull Soc Fr Dermatol Syphiligr. 1910; 17, 32.
-
World Health Organization Collaborating Centre for Oral Precancerous lesions Definition of leukoplakia and related lesions: an aid to studies on oral precancer. Oral Surg Oral Med Oral Pathol. 1978; 46(4), 518–39.
-
Pindborg JJ, Reichart PA, Smith CJ, Van der Waal I & World Health Organization. Histological typing of cancer and precancer of the oral mucosa. Berlin: Springer, 1997.
-
Van der Waal I. Oral potentially malignant disorders: Is malignant transformation predictable and preventable? Med Oral Patol Oral Cir Bucal. 2014; 19(4), e386–e390.
-
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010; 127(12), 2893–917.
-
Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2010, National Cancer Institute. [serial on the Internet]. June 2013 [cited: 1st May 2020]; Available from: https://seer.cancer.gov/archive/csr/1975_2010/.
-
Gupta PC, Mehta FS, Daftary DK, Pindborg JJ, Bhonsle RB, Jalnawalla PN, Sinor PN, Pitkar VK, Murti PR, Irani RR, Shah HT, Kadam PM, Iyer KS, Iyer HM, Hedge AK, Chandrashekar GK, Shiroff BC, Sahiar BE, Mehta MN. Incidence rates of oral cancer and natural history of oral precancerous lesions in a 10-year follow-up study of Indian villagers. Community Dent Oral Epidemiol. 1980; 8(6), 287–333.
-
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2008. CA Cancer J Clin. 2008; 58(2), 71–96.
-
Luryi AL, Chen MM, Mehra S, Roman SA, Sosa JA, Judson BL. Treatment Factors Associated With Survival in Early-Stage Oral Cavity Cancer: Analysis of 6830 Cases From the National Cancer Data Base. JAMA Otolaryngol Head Neck Surg. 2015; 141(7), 593–8.
-
Eisen D. The clinical features, malignant potential, and systemic associations of oral lichen planus: a study of 723 patients. J Am Acad Dermatol. 2002; 46(2), 207-14.
-
Lanfranchi-Tizeira H, Aguas SC, Sano SN. Transformación maligna del Líquen Plano Bucalatípico: Análisis de 32 casos. Med Oral. 2003; 8, 2-9.
-
Xue JL, Fan M-W, Wang S-Z, Chen X-M, Li Y, Wang L. A clinical study of 674 patients with oral lichen planus in China. J Oral Pathol Med. 2005; 34(8), 467-72.
-
Van der Meij EH, Mast H, van der Waal I. The possible premalignant character of oral lichen planus and oral lichenoid lesions: A prospective five-year follow-up study of 192 patients. Oral Oncology. 2007; 43(8), 742-8.
-
Van der Meij EH, Schepman KP, Smeele LE, van der Wal JE, Bezemer PD, van der Waal I. A review of the recent literature regarding malignant transformation of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999; 88(3), 307–10.
-
Epstein JB, Wan LS, Gorsky M, Zhang L. Oral lichen planus: progress in understanding its malignant potential and implications for clinical management. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003; 96(1), 32-7.
-
Van Der Meij EH, Schepman KP, Van Der Waal I. The possible premalignant character of oral lichen planus and oral lichenoid lesions: A prospective study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003; 96(2), 164-71.
-
Liu Y, Messadi DV, Wu H, Hu S. Oral lichen planus is a unique disease model for studying chronic inflammation and oral cancer. Medical Hypotheses. 2010; 75(6), 492–4.
-
Gonzalez-Moles MA, Scully C, Gil-Montoya JA. Oral lichen planus: controversies surrounding malignant transformation. Oral Diseases. 2008; 14(3), 229–43.
-
Mignogna MD, Fedele S, Lo Russo L, Lo Muzio L, Bucci E. Immune activation and chronic inflammation as the cause of malignancy in oral lichen planus: is there any evidence? Oral Oncology. 2004; 40(2), 120–30.
-
Kawanishi S, Hiraku Y, Pinlaor S, Ma N. Oxidative and nitrative DNA damage in animals and patients with inflammatory diseases in relation to inflammation-related carcinogenesis. Biological Chemistry. 2006; 387(4), 365–72.
-
Ergun S, Troşala ŞC, Warnakulasuriya S, Ozel S, Onal AE, Ofluoglu D, Guven Y, Tanyeri H. Evaluation of oxidative stress and antioxidant profile in patients with oral lichen planus. J Oral Pathol Med. 2011; 40(4), 286–93.
-
Valente G, Pagano M, Carrozzo M, Carbone M, Bobba V, Palestro G, Gandolfo S. Sequential immunohistochemical p53 expression in biopsies of oral lichen planus undergoing malignant evolution. J Oral Pathol Med. 2001; 30(3), 135-40.
-
Lee JJ, Kuo MJ, Cheng J, Chiang CP, Jeng JH, Chang HH, Kuo YS, Lan WH, Kok SH. Higher expressions of p53 and proliferating cell nuclear antigen (PCNA) in atrophic oral lichen planus and patients with areca quid chewing. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005; 99(4), 471-8.
-
Bascones C, Gonzalez-Moles MA, Esparza G, Bravo M, Acevedo A, Gil-Montoya JA, Bascones A. Apoptosis and cell cycle arrest in oral lichen planus Hypothesis on their possible influence on its malignant transformation. Arch Oral Biol. 2005; 50(10), 873-81.
-
Conway DI, Petticrew M, Marlborough H, Berthiller J, Hashibe M, Macpherson LM. Socioeconomic inequalities and oral cancer risk: a systematic review and meta-analysis of case-control studies. Int J Cancer. 2008; 122(12), 2811–9.
-
Leemans CR, Braakhuis BJ, Brakenhoff RH. The molecular biology of head and neck cancer. Nat Rev Cancer. 2011; 11(1), 9–22.
-
Bermejo-Fenoll A, Sanchez-Siles M, López-Jornet P, Camacho-Alonso F, Salazar-Sanchez N. Premalignant nature of oral lichen planus. A retrospective study of 550 oral lichen planus patients from south-eastern Spain. Oral Oncol. 2009; 45(8), e54–e56.
-
Goon PK, Stanley MA, Ebmeyer J, Steinstrasser L, Upile T, Jerjes W, Bernal-Sprekelsen M, Gorner M, Sudhoff HH. HPV and head and neck cancer: A descriptive update. Head Neck Oncol. 2009; 1, 36.
-
D’Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, Westra WH, Gillison ML. Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med. 2007; 356(19), 1944–56.
-
Sahebjamiee M, Sand L, Karimi S, Biettolahi JM, Jabalameli F, Jalouli J. Prevalence of human papillomavirus in oral lichen planus in an Iranian cohort. J Oral Maxillofac Pathol. 2015; 19(2), 170–4.
-
Sand L, Jalouli J. Viruses and oral cancer. Is there a link? Microbes Infect. 2014; 16(5), 371–80.
-
Boorghani M, Gholizadeh N, Zenouz A, Vatankhah M, Mehdipour M. Oral Lichen Planus: Clinical Features, Etiology, Treatment and Management; A Review of Literature. J Dent Res Dent Clin Dent Prospect. 2010; 4(1), 3–9.
-
Keenan AV, Ferraiolo D. Insufficient evidence for effectiveness of any treatment for oral lichen planus. Evidence-Based Dentistry. 2011; 12(3), 85–6.
-
Mattsson U, Jontell M, Holmstrup P. Oral lichen planus and malignant transformation: is a recall of patients justified? Crit Rev Oral Biol Med. 2002; 13(5), 390–6.
-
Sankaranarayanan R, Ramadas K, Thomas G, Muwonge R, Thara S, Mathew B, Rajan B. Trivandum Oral Cancer Screening Study Group. Effect of screening on oral cancer mortality in Kerala, India: a cluster-randomised controlled trial. Lancet. 2005; 365(9475), 1927–33.
-
Steele TO, Meyers A. Early detection of premalignant lesions and oral cancer. Otolaryngol Clin North Am. 2011; 44(1), 221–9.
-
Kalavrezos N, Scully C. Mouth Cancer for Clinicians Part 6: Potentially Malignant Disorders. Dent Update. 2015; 42(9), 866-8, 871-4, 877.
-
Mignogna MD, Lo Muzio L, Lo Russo L, Fedele S, Ruoppo E, Bucci E. Metastases in small thickness oral squamous cell carcinoma arising in oral lichen planus. Med Oncol. 2001; 18(2), 159-63.